A New Breakthrough in Prostate Cancer Precision Diagnosis
For prostate cancer patients who have undergone radical prostatectomy, monitoring prostate-specific antigen (PSA) levels is crucial for detecting cancer recurrence. When PSA rises again (biochemical recurrence) but conventional imaging fails to locate the lesions, precisely identifying the recurrence sites becomes a significant clinical challenge. Recently, a study called Co-PSMA has brought exciting news.
Head-to-Head Comparison Shows Promising Results for Novel Imaging Agent
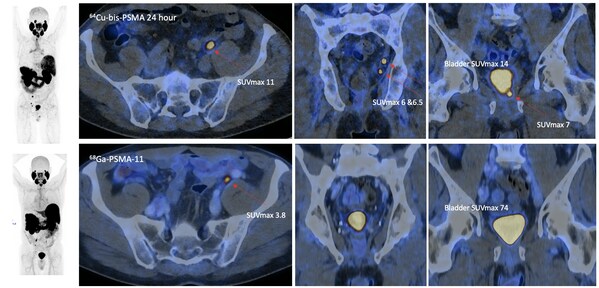
This clinical study, supported by Australian company Clarity Pharmaceuticals, was presented orally at the 2026 European Association of Urology (EAU) Congress, and its data has been accepted for publication in the top-tier journal European Urology. The study compared the diagnostic performance of the novel imaging agent 64Cu-SAR-bisPSMA with the current standard agent 68Ga-PSMA-11 in 50 patients with biochemical recurrence and low PSA levels (0.2 – 0.75 ng/mL).
Key Findings: More Lesions, Higher Detection Rate
The results demonstrated a clear superiority for 64Cu-SAR-bisPSMA at 24-hour imaging:
- Doubled Lesion Detection: 64Cu-SAR-bisPSMA identified 63 lesions in total, compared to only 24 lesions detected by 68Ga-PSMA-11.
- Significantly Higher Positive Scan Rate: 78% of patients (39/50) had a positive scan with the new agent, versus only 36% (18/50) with the standard agent.
- Improved Diagnostic Accuracy: 64Cu-SAR-bisPSMA showed a higher true positive rate (71%) and a lower false negative rate (21%) compared to 68Ga-PSMA-11 (29% and 65%, respectively).
The advantage was most pronounced in detecting lesions in the prostatic bed (local recurrence) and lymph nodes.
Direct Impact on Clinical Decision-Making
More precise diagnosis directly altered patient treatment pathways. The study found that after "next-day imaging" with 64Cu-SAR-bisPSMA, the treatment plan changed for 44% of participants (22/50), with the majority shifting from active surveillance to targeted radiotherapy. This means more patients have the opportunity to receive effective curative salvage therapy at an earlier stage of recurrence.
Future Outlook
Data from the Co-PSMA study, along with results from other related clinical trials, are intended to be submitted to the U.S. Food and Drug Administration (FDA) for market authorization of 64Cu-SAR-bisPSMA in patients with biochemical recurrence of prostate cancer. This advancement marks a significant step forward in precision imaging for prostate cancer, potentially helping more patients receive timely and effective intervention at an early stage of recurrence.